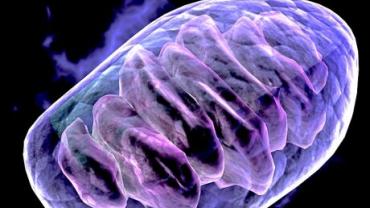
Mitochondrial insufficiency and dysfunction are contributing factors in many of the debilitating health conditions that burden individual families and national healthcare resources alike. Malfunctioning of these energy-producing organelles has been shown to cause or exacerbate the pathology and progression of cancer Alzheimer’s disease Parkinson’s disease multiple sclerosis and more. With this in mind pharmaceutical drugs nutritional supplements dietary interventions and lifestyle practices that can boost mitochondrial density and efficiency are at the forefront of cutting-edge academic and clinical research along with a growing interest among laypeople looking to optimize their health and longevity.
Intermittent fasting is an effective yet simple—and free!—way for patients to start. Autophagy and beneficial cellular changes induced by the hormetic stress of intermittent fasting cascade through multiple organ and tissue systems with potential benefits that are difficult to fully catalog. Physical exercise is another way to induce mitochondrial biogenesis. Diets rich in antioxidants anti-inflammatory compounds and long-chain omega-3 fatty acids may help reduce oxidative stress and inflammation and help support healthy plasma membrane synthesis including those of the double-membraned mitochondria. Medium chain triglycerides (MCTs) owing to their unique conversion to ketone bodies may help to nourish cells whose mitochondria are not able to metabolize glucose effectively such as occurs in Alzheimer’s. MCTs have also been used to provide nourishment to healthy cells and reduce cancer-induced cachexia owing in part to cancer cells’ near-total dependence on glucose metabolism due to mitochondrial dysfunction.
With all this in mind interest in and demand for other interventions that might boost mitochondrial efficiency are growing. One such intervention is supplementation with the compound PQQ. PQQ—short for pyrroloquinoline quinone—is a cofactor for bacterial redox reactions and is a signaling agent for proteins kinases involved in differentiation of mammalian cells. The high redox recycling ability of PQQ may give it a pharmacological role in protecting against neurodegeneration and cancer. (As a redox agent PQQ is extremely stable and can participate in hundreds more reactions than other antioxidants such as ascorbic acid quercetin and epicatechin.)
Mammals do not synthesize PQQ and while it is not currently classified as a vitamin or otherwise essential nutrient PQQ is believed to be important in mammalian nutrition and may even be required for normal growth and development. In humans PQQ is typically found in miniscule concentrations: nanograms per gram. Like any such substance however—be it a vitamin mineral or a “non-essential” nutrient—certain health conditions may benefit from higher concentrations increased via supplementation with levels above those typically obtained from foods.
Among a selection of vegetables fruits tea alcoholic beverages and soy products tested for PQQ content PQQ was found in all samples ranging from 3.7 - 61 ng/g or ng/ml. The highest concentration was found in natto a fermented soy food that is also the richest source for another underappreciated nutrient vitamin K2. Parsley papaya green peppers and oolong and green teas were found to be good sources of PQQ. Animal foods including eggs and dairy have been shown to contain large amounts of PQQ but the detection and assay methods used have been called into question. Since mammalian cells cannot synthesize PQQ researchers believe the PQQ found in human tissue is likely derived from the diet as well as originating from enteric bacterial production.
Rat studies show impressive efficacy for PQQ in stimulating mitochondrial biogenesis. Rat hepatocytes incubated with PQQ at 10–30 μM for 24–48 h exhibited “increased citrate synthase and cytochrome c oxidase activity Mitotracker staining mitochondrial DNA content and cellular oxygen respiration. The induction of this process occurred through the activation of cAMP response element-binding protein (CREB) and peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α) a pathway known to regulate mitochondrial biogenesis.” In vivo studies in rats also show beneficial effects from dietary supplementation with PQQ (2mg PQQ/kg of food). These include decreased plasma triglycerides increased energy expenditure (correlated with hepatic mitochondrial content) and improved tolerance to cardiac ischemia/reperfusion. Experimental models of stroke and spinal cord injury show that PQQ attenuates neuronal cell death in part through PQQ protecting N-methyl-d-aspartic acid (NMDA) receptors. Rat models of Parkinson’s disease show that PQQ supplementation decreases neuronal loss increases reactive oxygen species scavenging ability and offers neuroprotection through additional mechanisms.
Much of the research on PQQ to date has been conducted in animals but there is a growing body of work supporting its use in humans with no evidence of acute side effects or overt toxicity from amounts as high as 60 mg per day. Oral PQQ supplementation does increase indicators of mitochondrial efficiency in healthy humans. More work needs to be done in investigating the efficacy of PQQ for conditions known to have underpinnings in mitochondrial dysfunction but evidence so far is quite compelling for PQQ’s role in helping to reduce age-related oxidative stress and rejuvenating healthy mitochondria.